
CS1 maint: Multiple names: authors list ( link) CS1 maint: Multiple names: authors list ( link) CS1 maint: Extra text: authors list ( link) CS1 maint: Extra text ( link) Perry's Chemical Engineers' Handbook (7th Edition ed.).
DEPRIESTER CHART ISOBUTANE HOW TO
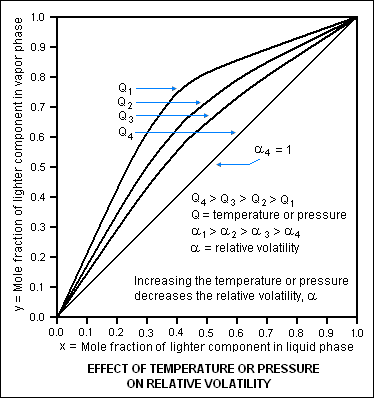
By convention, relative volatility is usually denoted as α values are widely used in the design of large-scale distillation columns for distilling multi-component mixtures in oil refineries, petrochemical and chemical plants, natural gas processing plants and other industries. Describes how to use an interactive simulation that demonstrates how to read vapor-liquid equilibrium ratios from a DePriester chart. Use the DePriester chart to generate the temperature-composition diagram for isobutane and propane at 1000 kPa. Taking into consideration isobutane consumption in MTBE and butyl rubber production, it becomes clear, that isobutane is a deficit product and must be replenished by normal butane isomerization. In effect, it indicates the ease or difficulty of using distillation to separate the more volatile components from the less volatile components in a mixture. This quantity is widely used in designing large industrial distillation processes. Prog.Relative volatility is a measure comparing the vapor pressures of the components in a liquid mixture of chemicals.

Tham, University of Newcastle upon Tyne (scroll down to Relative Volatility) Halvorsen and Sigurd Skogestad, Norwegian University of Science and Technology (scroll down to: 2.2.3 K-values and Relative Volatility) Values are widely used in the design of large-scale distillation columns for distilling multi-component mixtures in oil refineries, petrochemical and chemical plants, natural gas processing plants and other industries. Thus, for the distillation of any multi-component mixture, the relative volatility is often defined as In that context, a lighter component means a component with a lower boiling point (or a higher vapor pressure) and a heavier component means a component with a higher boiling point (or a lower vapor pressure). The designer would designate the key components governing the separation design to be propane as the so-called light key (LK) and isobutane as the so-called heavy key (HK). Such a distillation column is typically called a depropanizer. A bottoms fraction containing predominantly the less volatile components ranging from isobutane (having four carbon atoms) to decanes (ten carbon atoms).An overhead fraction containing predominantly the more volatile components ranging from methane (having one carbon atom) to propane (having three carbon atoms).For distilling such a mixture, the distillation column may be designed (for example) to produce: For example, some intermediate products in an oil refinery are multi-component liquid mixtures that may contain the alkane, alkene and alkyne hydrocarbons ranging from methane having one carbon atom to decanes having ten carbon atoms. When a multi-component mixture is distilled, the overhead fraction and the bottoms fraction typically contain much more than one or two components. Typically, the overhead fraction from the distillation column consists predominantly of the more volatile component and some small amount of the less volatile component and the bottoms fraction consists predominantly of the less volatile component and some small amount of the more volatile component.Ī liquid mixture containing many components is called a multi-component mixture. Perry and Green 5, and Smith and Van Ness 6 provide DePriester charts in alternative. When a binary mixture is distilled, complete separation of the two components is rarely achieved. Table 4.2 Constants for fit to Kvalues using Equation 4.2.
DEPRIESTER CHART ISOBUTANE TV
Xg 0.089, xc 0.136, xp 0.687 Pressure, psia Methane Ethylene Ethane Tv (a) Propylene 1 Propane Isobutane -Butane Isopentane - Pentane -Hexane 10 3 - Heptane n-Octane -Nonane g Temperature, 'F Pressure. Increases above 1, separation by distillation becomes progressively easier.Ī liquid mixture containing two components is called a binary mixture. I provided the DePriester chart for more information of the K-value.
